  
In this article, the principle will not be mentioned only the results will be presented, as far as they have been determined in my experiments. The study of the relative weights of the particles is, to my knowledge, completely new I have recently continued this review with remarkable success. He maintained the computations in the months that followed. The first atomic calculations in the document appear to have been entered on September 6, 1803. We know this because of the notepad discovered in his lab (the notebook itself was destroyed by an airstrike in 1944, but the photostatic copy of the important pages was separately printed in 1896). However, as the excerpt below shows, he mentioned nothing to the audience about how he arrived at these figures. These were the six atom weights from his first article that he read to the audience on the stage in October 1803. (We now know that this ratio is eight to nine.) He applied the same process to carbon, nitrogen, sulfur, and phosphorus compositions. In summary, if the hydrogen atom is the H1 weight unit and the water molecule is HO, and if it is seven-eighths oxygen, then it should be O=7. Therefore, the oxygen atom must have been 7/8 of the weight of the water molecule. Weight analysis of the water at that time indicated that it was composed of 7/8 oxygen and 1/8 hydrogen. The second step was to analyze the composition (or make use of the analysis of other chemists). In today’s language, Dalton thought the water formula was HO at that time. He thought that the most likely answer would be to connect a single oxygen atom to a single hydrogen atom to form water. 
He knew that liquid water was composed of gaseous hydrogen and oxygen elements, but what would a single molecule of this substance look like? In Dalton’s ingenious method, the first step was to visualize how simple compounds such as water are formed if they could be observed at the invisible level of the particles. For this purpose, he assigned the weight of 1 to the lightest atom, namely hydrogen, and tried to determine the weight of each element’s atoms relative to 1. However, he thought that there could be a way to find their relative weight. Because they were too small to be measured and detected. It was not possible to measure the atoms of various elements directly. He believed that the only way to fully understand these substances was to first deduce how heavy the absolute particles of the chemicals were. John Dalton: Heavy Elements Table of atomic symbols and assumed relative weights that Dalton used in his classes in 1806-1807ĭue to his scientific interest, John Dalton turned to a more general examination of mixed gases and water-dissolved gases. Keeping his conversations lean, away from controversy or falsehood, Dalton continued his research quietly, with intellectual courage and mental brilliance, which he never emphasized. However, he had a very lively scientific imagination. Although he was not highly blessed in mathematics, his mind was extremely prone to numbers and mathematical concepts, which he applied to nature through intuition. He never married but had a certain number of close friends who deeply appreciated his gentle personality and simplistic and philosophical approach as a Quaker. John Dalton lived a quiet life in this vibrant and rising British industrial city. He was assigned a chassis study room and a laboratory in the lodgings. He made his living by giving private math and chemistry lessons. When he left New College, he was elected secretary of the Literary and Philosophical Society. Dalton resigned from his position but stayed in Manchester. The school was “ opposed to the Anglican Church,” and in 1800 it started to face financial difficulties and failed to pay wages. In the same year, he accepted a teaching position in nature philosophy at New College in Manchester. In 1793, his first book, Meteorological Observations and Essays, was published. John Dalton’s favorite pursuit at that time was meteorology he specialized in this field later and continued his research with passion until the end of his life. Three years later, he joined his older brother, who runs a boarding school in the nearby town of Kendal. In his spare time, he continued his education by studying classical and modern languages, mathematics, and the natural sciences. Dalton started teaching at the village school at the age of 12. Even among their peers, the Cumberland Quakers stood out for the importance they placed on education and mental pursuits. 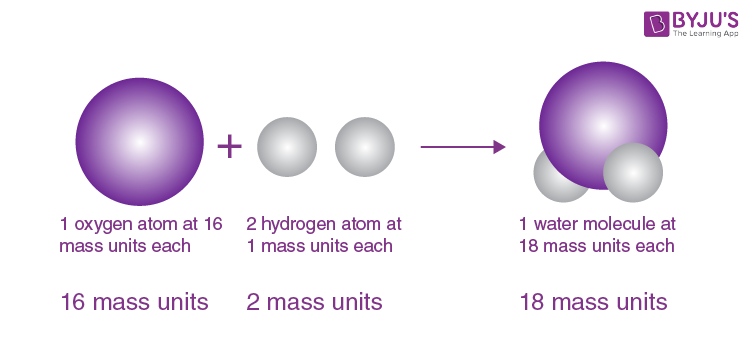
He spent his first years working on the family’s small farm, but on the other hand, he had a passion for education and became self-educated with the help of local elite Quakers. Dalton’s father was Cockermouth, a poor weaver who lived in Cumberland, on the northwest tip of England. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
